 中文
中文
|
No. |
Strain Type |
Name |
Source |
Licensing Fee |
| 1 | E.coli |
Stbl3 Competent Cells |
Thermo Fisther | R&D to IND:free |
| 2 |
DH5α Competent Cells |
Thermo Fisther | ||
| 3 |
BL21 (DE3) Competent Cells |
Merck | ||
| 4 |
Rosetta (DE3) Competent Cells |
Merck | ||
| 5 | Yeast |
X-33 Pichia pastoris |
Thermo Fisther | After BLA:TBD |
| 6 |
GS115 Pichia pastoris |
Thermo Fisther |
Key Advantages
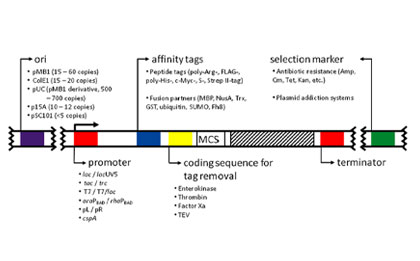
Multiple plasmid element designs to optimize product expression in engineered strains
A diverse engineered strain library to meet the needs of different products
A rapid and efficient screening platform
Low project cost
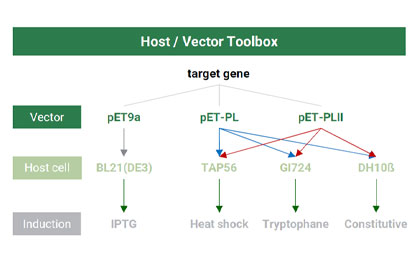
Screening for the optimal production cell line and the most cost-effective induction mode
High-yield expression of target products
Flexible operability