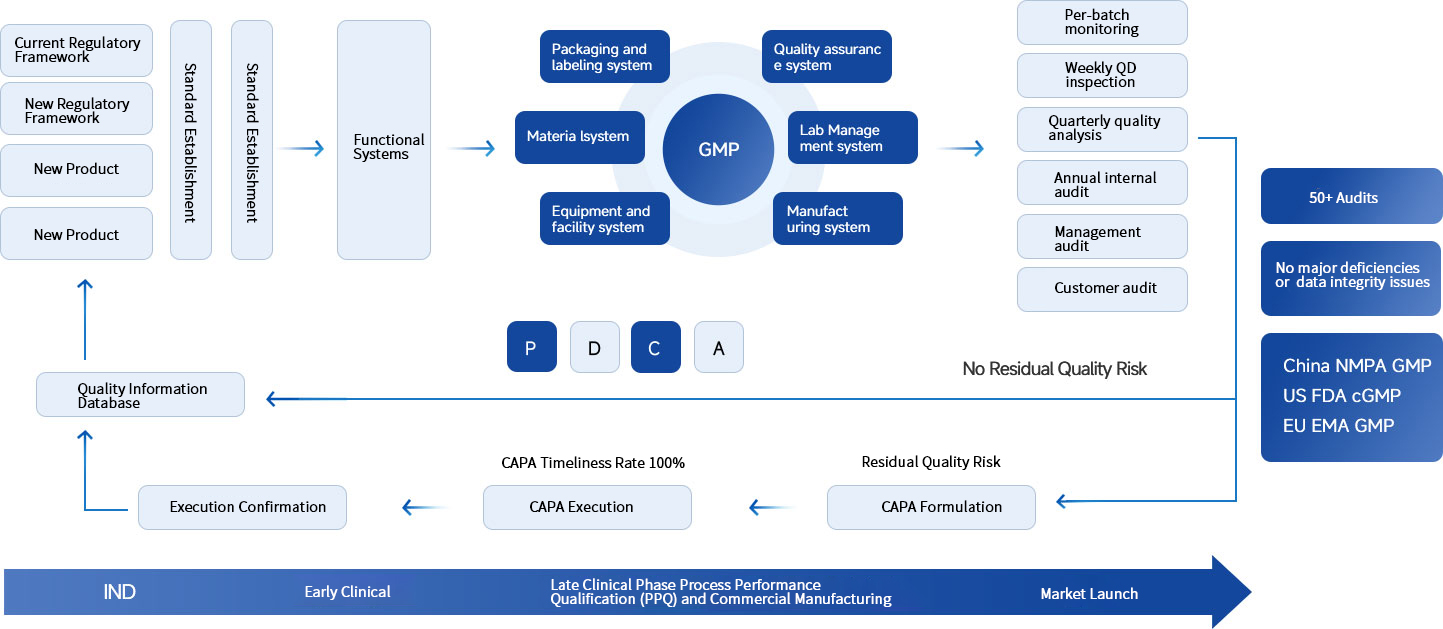
Analytical method transfer, validation/verification (physicochemical, biochemical, cellular, and microbiological);
Release testing for in-process intermediates, bulk drug substances, and finished drug products;
Sampling and testing for utility systems: water, environment, gases, etc.
Release testing for drug substances and packaging materials.

 中文
中文