I. Project Background
The target molecule is an IgM-form neutralizing antibody against SARS-CoV-2. It needs to be developed as a multi-dose nasal spray liquid formulation to provide mucosal protection. The project timeline is tight, requiring completion of the entire process from R&D to scale-up within an extremely short timeframe.
II. Technical Challenges
IgM antibodies typically exhibit low expression levels, and enhancing expression is challenging. There is a risk of expressing a high proportion of hexamer impurities. Difficulties include virus filtration and removal of aggregates. The intended use under ambient conditions, combined with the nasal spray device, poses significant challenges to formulation stability. Analytical characterization of IgM antibodies is also challenging.
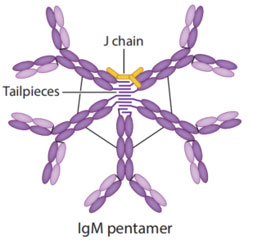
III. IgMax® Vector Platform Addressing Expression Challenges
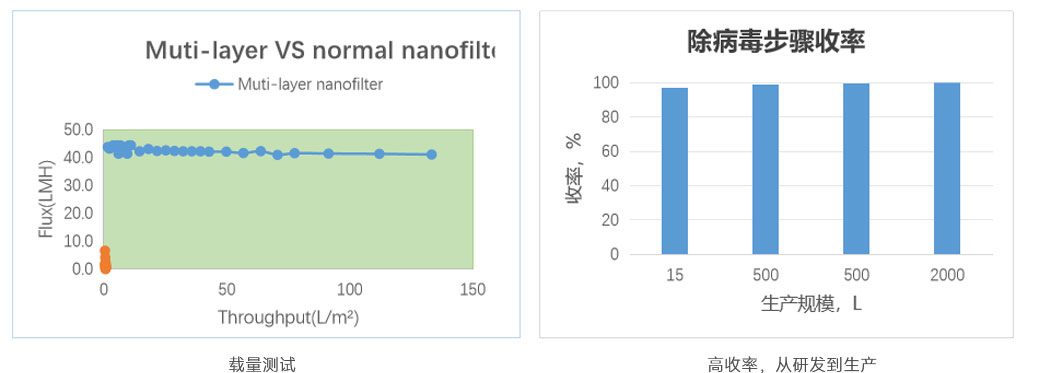
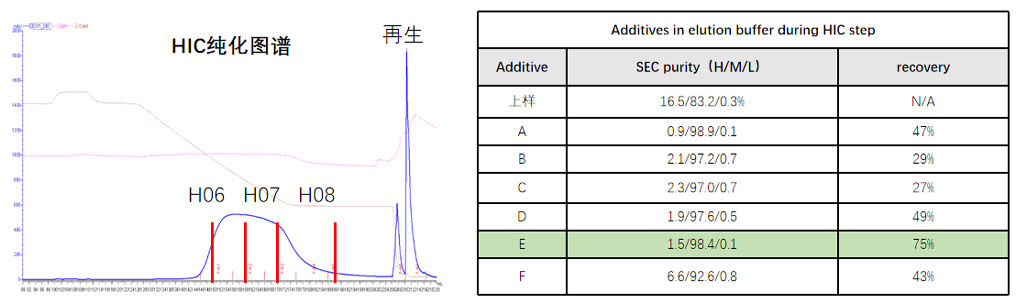
IV. Downstream Process Optimization Addressing Process Challenges